Problems due to limescale and water hardness
- Hard water reduces the oxidizing power of Lotus Clean Shock.
- Lotus Clean Basic does not behave optimally with hard water.
- Limestone is present in the water in dissolved form, chemically bound to carbon dioxide. The harder the water, the more carbon dioxide there is in the water.
- When the water is heated and swirled, the carbon dioxide from >> pH value of the water rises >> the dissolved lime turns into limestone >> the water can become cloudy.
- The limestone can settle in the approx. 150 m² of pipe walls.
- Bacteria use limestone as a source of food.
- Limestone surface is rough and oils and cosmetics can settle. Thus a biofilm forms very fast in the pipelines.
This is how biofilm is formed in the whirlpool
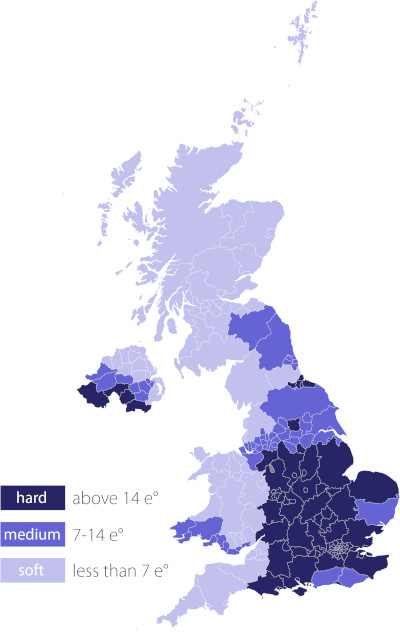
Water hardness
The harder the water is (above 14 °e), the more important it is to proceed correctly to prevent limescale deposits from forming. The local water hardness can be requested from the water suppliers.
Products against lime and water hardness


Lotus Clean pH-Minus liquid
To avoid lime precipitation, keep the pH value of the water at 6.8 to 7.2, i.e. rather in the slightly acidic range. Especially suitable is our pH-Minus liquid:
- It has a triple effect against limescale
- It converts lime ions into water-soluble salts
- It dissolves existing lime from the pipelines
- It acts as a complexing agent and removes metal ions from the water
Lotus Clean Calc Down Pad
The Lotus Clean Calc Down Pad contains a special mineral which is charged with sodium ions. In water, these minerals release sodium ions and absorb calcium ions. This makes the water softer.
Important for a fast and effective softening of the water is the good flow through the Calc Down Pads. It is best to place them in the filter chamber. For whirlpools without a filter chamber, the Calc Down Pads should be placed directly on the filter. It can be useful to increase the filter running time during softening..
After use, the bags must be placed in distilled water with at least 30% salt content. This regenerates the Calc Down Pads.


Total Hardness Test Kit
The Total Hardness Test Kit is for measuring water hardness.
Lotus Clean Calcium Ex
Calcium Ex is a complexing agent. During the transformation of lime ions into limestone, the complexing agent "grabs" the lime molecule and combines with it. The molecular structure is changed in such a way that it can no longer cling to itself. The lime floats in the water and is filtered out.

The ph-value
The pH value indicates whether a water is alkaline, acidic or neutral. The scale ranges from 0 to 14, at 7 it's neutral.

The pH value is logarithmic
- A pH of 6 is ten times as acidic as a pH of 7 is.
- A pH value of 5 is 100 times as acidic.
- A pH value of 4 is 1000 times as acidic.
If the water is very hard, you should lower the pH value of the water to 6 for one week approx. every 8 weeks
- The water becomes acidic and dissolves existing limescale.
- Bathing remains harmless as the skin has a pH value of 5.5.
- The skin reacts rather sensitively to high pH values above 8.0.
The pH value should be between 6.8 and a maximum of 7.8
- High pH values can lead to green and cloudy water.
- Too low pH values can lead to corrosion.
- From a pH value of 8, lime precipitation can occur, which we absolutely want to avoid.
